Microglia orchestrate the central nervous system (CNS) response to injury and disease. Yet it remains unclear whether the microglial response to disease is beneficial or harmful. A contributing factor to this uncertainty is that it has been technically challenging to track microglia without also following peripheral macrophages, a cell population that differs in function and toxicity. The Plemel lab will use new tools to monitor and ablate microglia specifically to investigate their role in white matter regeneration, or remyelination, and degeneration.
One of the lab’s central research interests is Multiple Sclerosis (MS), which is an inflammatory disease with persisting neurodegeneration affecting 24-30 out of 10,000 Canadians. For most people with MS, the later stages are characterized by a continuing increase in disability and neurodegeneration, known as progressive MS. Currently, there are limited medications to slow MS progression, likely because of the lack of knowledge of progression mechanisms. In progressive MS, microglial activity measured by PET scans corresponds to the progression of disability; thus, microglia are thought to play an essential role in the neurodegenerative process. However, it is unclear whether microglia respond to injury or drive it. Moreover, if microglia are responding to their environment, is their behaviour beneficial, harmful or both? The Plemel lab is exploring the response of microglia during white matter degeneration to help untangle the complex microglial response during progressive MS.
In a related project, my lab will investigate how the regeneration of lost myelin is regulated by microglia. MS is characterized by white matter lesions where myelin is damaged. In these lesions, many axons survive, and in some people, these axons are even remyelinated. However, for the majority of people with MS, remyelination is limited. Remyelination may be a way to treat people with MS because those individuals who have more vigorous remyelination also tend to have less disability.
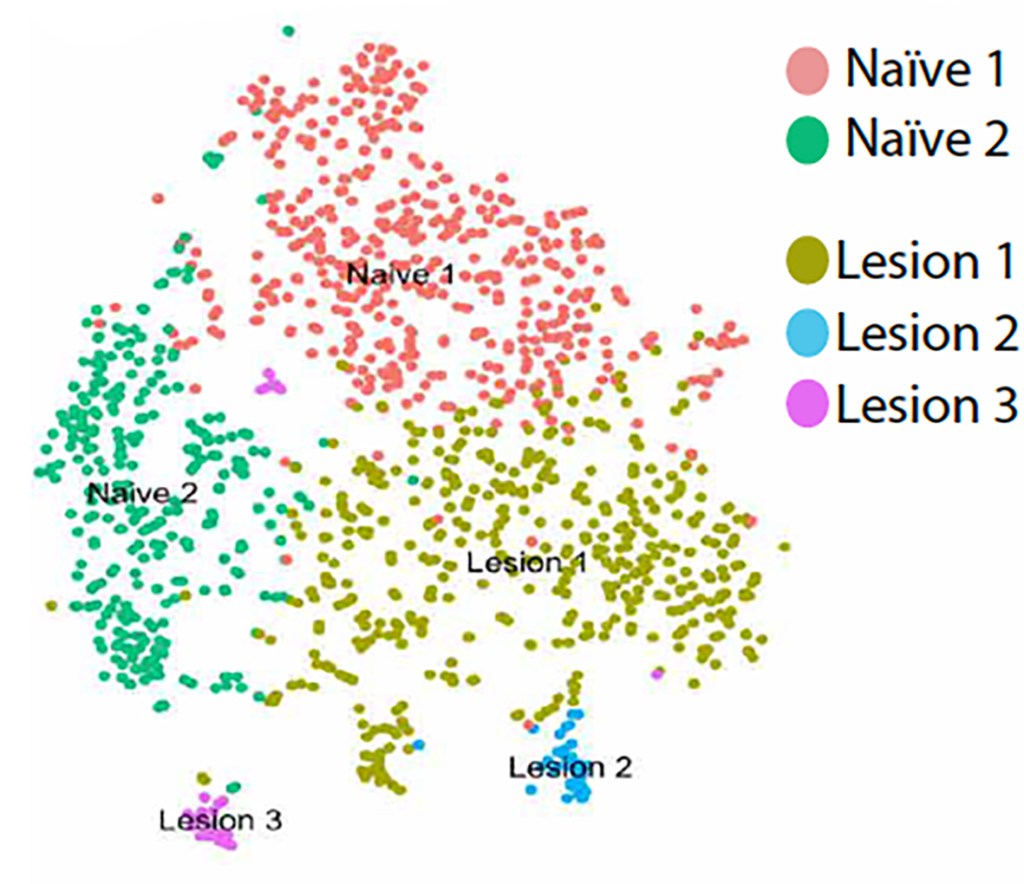
Indeed, several medicines targeting remyelination are now entering clinical trials to see if they can improve outcomes in people with MS. These medicines work by encouraging the production of new oligodendrocytes. While this is an important strategy, these therapies do not foster the local environment where remyelination takes place, which is regulated by immune cells such as microglia and macrophages. Recently, the Plemel lab and its collaborators identified new populations of microglia during an early stage of remyelination with single-cell RNA sequencing. The Plemel lab is now investigating the role of microglia and CNS-infiltrating macrophages during remyelination. Our overarching goal is to understand the characteristics of regeneration-associated microglia so that this phenotype can be enhanced as a way to treat MS and other CNS diseases.